4 He thought that chemistry could be seen as just another version of applied math. We're officially masters of molecular masses.īrain SnackJeremias Benjamin Richter (1762-1807) (not to be confused with Andy Richter) was the first guy to lay down the principles of stoichiometry. 
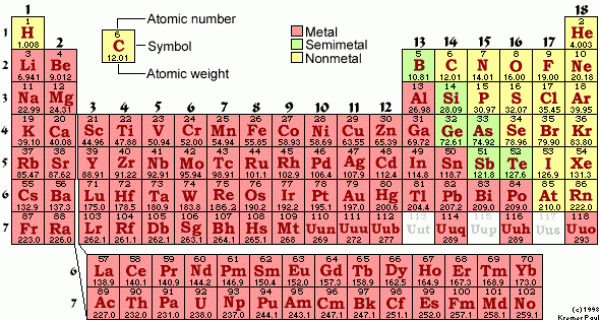
We need to arrange the conversion factor in whichever way will allow us to do that. Remember, if a unit appears above and below the fraction line they will cancel each other out. We just need to use the conversion factor that will give us the right units in the end. What gives? Why do we need two conversion factors? We don't. Note: we multiply by 4 because there are 4 hydrogen atoms in each CH 4. Molar mass of CH 4 = atomic mass carbon + 4(atomic mass of hydrogen) We need to collect the atomic masses of carbon and hydrogen from the periodic table and then calculate the molecular mass of CH 4 by simple addition. Once again, the periodic table is your BFF. This means we need a conversion factor between grams and moles. How many moles of CH 4 are present in 4.83 g of CH 4?įirst of all, we are given grams of CH 4 and asked to solve for moles of CH 4. Who knew we were drinking so many molecules? Sample ProblemOne of the main components of natural gas is methane (CH 4). Think about that next time you down a glass of water. That means every 1.2 tablespoons of water contains 6.023 x 10¬ 23H 2O molecules. Those 18.02 grams represent one mole of water. Roughly 1.2 tablespoons of water is equal to 18.02 grams. That means 1 mole of water is equal to 18.02 g. :max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg)
Lucky for us (thanks again to the chemistry gnomes), its molar mass is 18.02 g. We just calculated the molecular mass of water to be 18.02 amu. The molar mass of a compound (in grams) is numerically equal to its molecular mass (in amu). We don't even need a calculator to compute between molecular mass and molar mass…the values are numerically the same. Just as the name suggests the molar mass is the mass (in grams) of one mole of a compound. Molecular mass H 2O = 2(1.008 amu) + 16.00 amu = 18.02 amuįrom the molecular mass we can determine the molar mass of a molecule. We know, we heart the periodic table, too. Where do we find the atomic mass of H and O? Whip out your favorite periodic table. Molecular mass H 2O = 2(atomic mass of H) + atomic mass of O What's the molecular mass of water (H 2O)? To calculate the molecular mass we add together the atomic mass of two hydrogen atoms and one oxygen atom: The molecular mass (sometimes called the molecular weight) is just the sum of the atomic masses in a molecule. We don't want to offend all the atoms out there.) If we know the atomic masses of individual atoms, we can easily calculate the mass of a molecule. Atoms are so small, however, that even 500 atoms are too small to see or measure by most common techniques.Now that we know all about atomic masses we can move on to bigger and better things…molecules. Sheets of printer paper are packaged in reams of 500, a seemingly large number. For example, cans of soda come in a six-pack, eggs are sold by the dozen (12), and pencils often come in a gross (12 dozen, or 144). Many familiar items are sold in numerical quantities with distinct names. The unit that provides this link is the mole (mol), from the Latin moles, meaning “pile” or “heap.” To analyze the transformations that occur between individual atoms or molecules in a chemical reaction, it is therefore essential for chemists to know how many atoms or molecules are contained in a measurable quantity in the laboratory-a given mass of sample. In the laboratory, for example, the masses of compounds and elements used by chemists typically range from milligrams to grams, while in industry, chemicals are bought and sold in kilograms and tons. Because the masses of individual atoms are so minuscule (on the order of 10 −23 g/atom), chemists do not measure the mass of individual atoms or molecules. The problem for Dalton and other early chemists was to discover the quantitative relationship between the number of atoms in a chemical substance and its mass. It also describes the law of multiple proportions, which states that the ratios of the masses of elements that form a series of compounds are small whole numbers. _3(PO_4)_2 = 310.177 \, amu \nonumber \]ĭalton’s theory that each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
